Calculate the enthalpy of formation of water, given that the bond energies of `H-H, O=O` and `O-... - YouTube
![Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1) Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1)](https://d10lpgp6xz60nq.cloudfront.net/web-thumb/69096014_web.png)
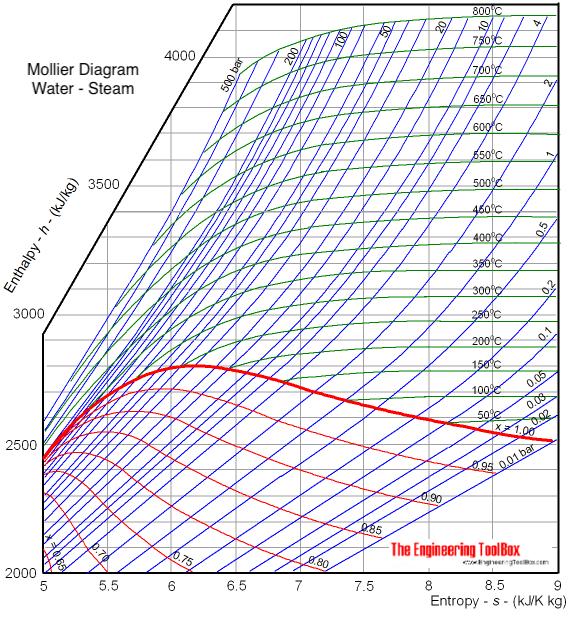
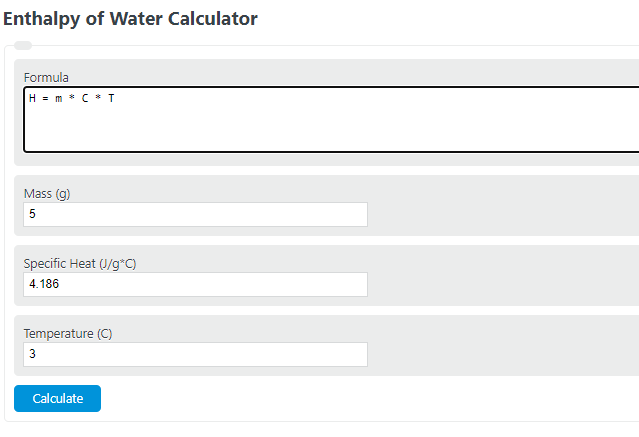
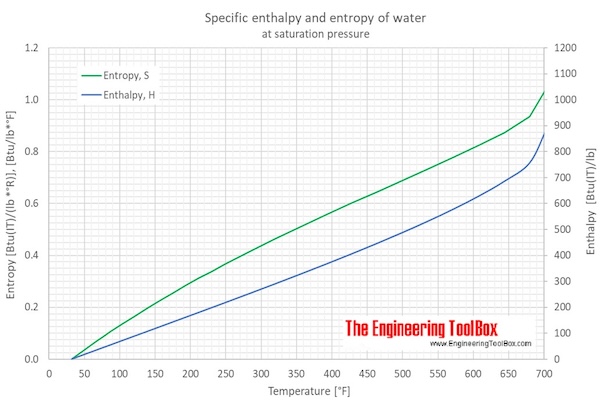
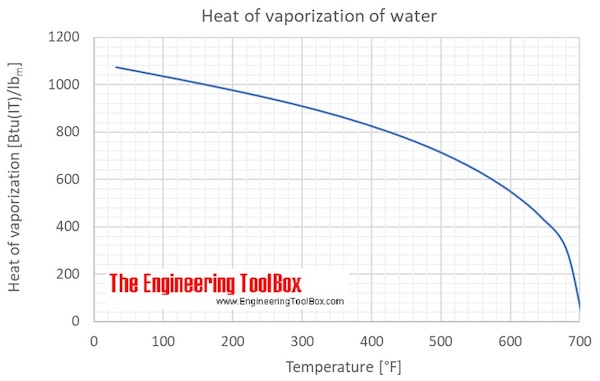
Calculate the enthalpy change of freezing of 1.0 mol of water at 10^(@)C to ice at -10^(@)C, Delta(fus)H=6.03 kJ mol^(-1) at 0^(@)C. C(P)[H(2)O(l)]=75.3 J mol^(-1) K^(-1) C(P)[H(2)O(s)]=36.8 J mol^(-1) K^(-1)

Question Video: Calculating the Enthalpy of Dilution of Sodium Hydroxide Using Enthalpy of Solution Values | Nagwa

Question Video: Determining a Standard Enthalpy Change Given the Standard Enthlapy of Fusion | Nagwa

Calculate the enthalpy change of freezing of `1.0` mol of water at `10^()C` to ice at `-10^()C, ... - YouTube

The enthalpy change for the formation of 3.6 kg water is _____ . `H_(2(g))+1/2 O_(2(g)) to H_2 O_ - YouTube

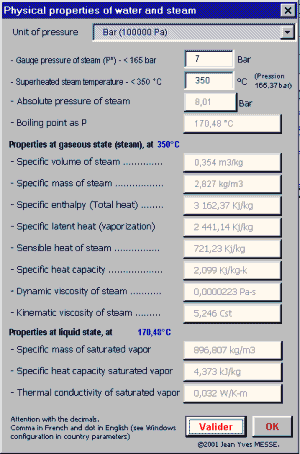
Calculate change in enthalpy when 2 moles of liquid water at 1 bar and `100^(@)` C is coverted into - YouTube

The enthalpy change for transition of liquid water to steam is 40.8 kJ `mol^(-1)` at 373K. - YouTube